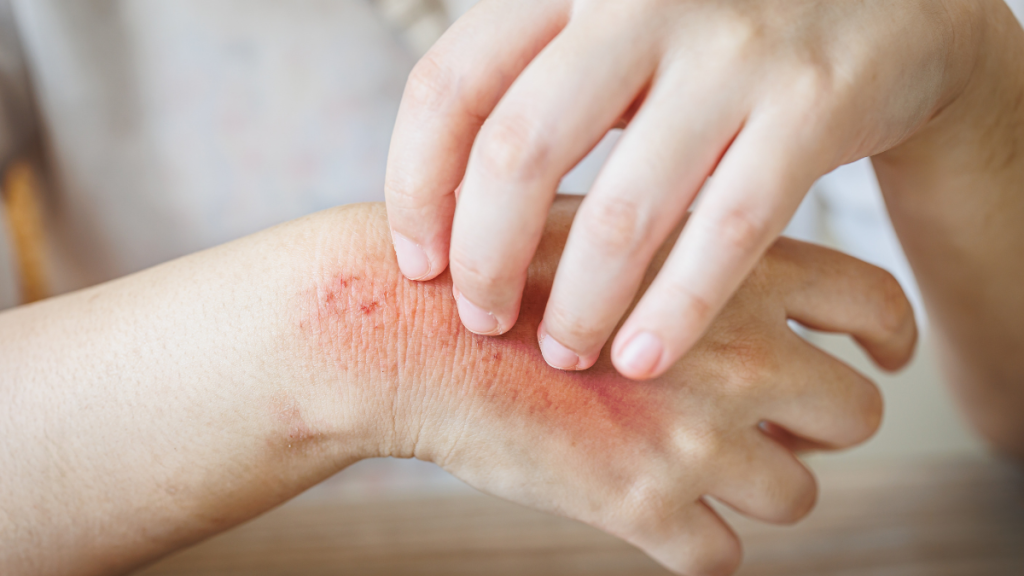
Summary
Health Canada authorized Nemluvio®, a first-in-class biologic for eczema and prurigo nodularis that specifically targets the biological cause of chronic itch. While national funding reviews are still underway, this approval provides a vital new option for patients who haven't found relief with standard creams.
A new era in dermatological care has arrived for Canadians suffering from chronic, debilitating itch. Health Canada authorized Nemluvio® (nemolizumab) for the treatment of moderate-to-severe atopic dermatitis (eczema) in patients aged 12 and older, as well as prurigo nodularis in adults.
Nemluvio® is a first-in-class biologic that specifically targets the neuro-immune pathway responsible for itch, providing a much-needed alternative for patients whose conditions are not adequately controlled by topical therapies.
Targeting the “Itch Cytokine”
Unlike traditional eczema treatments that focus primarily on skin inflammation, Nemluvio® is the first monoclonal antibody designed to block interleukin-31 (IL-31). Often referred to by scientists as the “itch cytokine,” IL-31 sends signals directly to the nervous system to trigger the urge to scratch.
By blocking this signal, Nemluvio® addresses the most burdensome symptom of these conditions: the relentless itch that leads to sleep deprivation, skin damage, and significant mental health strain.
“Nemolizumab’s ability to rapidly target itch makes it an exciting and much-needed advancement, offering patients relief where they need it most,” said Dr. Chih-ho Hong, Board-Certified Dermatologist, MD, FRCPC, Surrey, BC.
Proven Clinical Success
The Health Canada authorization was based on robust data from two major Phase III clinical programs:
- ARCADIA 1 & 2 (Atopic Dermatitis): These trials demonstrated that Nemluvio, administered every four weeks, significantly improved skin lesions and reduced itch in adults and adolescents.
- OLYMPIA 1 & 2 (Prurigo Nodularis): This program, the largest of its kind for this condition, showed that the treatment significantly improved itch and cleared skin nodules compared to a placebo.
Status in Canada: The Road to Access
While Nemluvio® is now authorized for sale in Canada, its widespread availability under public insurance is still in progress.
- Reimbursement Review: The drug is currently undergoing a cost-effectiveness review by Canada’s Drug Agency (CDA-AMC). This national review is a required step before the drug can be listed on federal, provincial, and territorial public drug plans.
- Cost & Funding: Because the review is ongoing, an official, widely available list price for patient use has not yet been finalized or publicly released. Until funding decisions are made by the provinces, Nemluvio® is not yet a regular benefit under most public insurance plans.
- Patient Support: Historically, specialty biologics are supported by manufacturer-led patient assistance programs. These programs often help patients navigate early access and insurance coverage during the reimbursement review phase.
Is It Right For You?
Nemluvio® is intended for those whose disease is not adequately controlled with topical prescription therapies. Because it is an injectable medication given once every four weeks, it offers a convenient schedule for those looking to move away from daily creams or more frequent injections.
Subscribe to The Health Insider newsletter for more evidence-based updates on Canadian medical breakthroughs.
~ Read more from The Health Insider ~
- Should You Take Berberine? What Health Canada and the Research Say
 Berberine is trending as “nature’s Ozempic”, but what does Health Canada actually approve it for, and is it safe to take with your medications?
Berberine is trending as “nature’s Ozempic”, but what does Health Canada actually approve it for, and is it safe to take with your medications? - How Exercise Actually Grows Your Brain
 Your muscles feed your brain! Learn how to trigger the natural chemicals that protect your memory and sharpen your daily focus.
Your muscles feed your brain! Learn how to trigger the natural chemicals that protect your memory and sharpen your daily focus. - What Is Hantavirus and Should Canadians Be Concerned?
 Hantavirus is rare but serious. Here’s what Canadians need to know about symptoms, how it spreads, and how to protect yourself.
Hantavirus is rare but serious. Here’s what Canadians need to know about symptoms, how it spreads, and how to protect yourself. - Radon Is the Leading Cause of Lung Cancer in Non-Smokers and Most Canadians Haven’t Tested for It
 One in five Canadian homes may have elevated radon but only 12% have ever tested. Here’s what you need to know.
One in five Canadian homes may have elevated radon but only 12% have ever tested. Here’s what you need to know. - Canada Just Approved a New Alzheimer’s Drug. Here’s What You Need to Know
 Health Canada has approved a new Alzheimer’s drug that targets the disease itself, not just symptoms. Here’s what Canadians need to know.
Health Canada has approved a new Alzheimer’s drug that targets the disease itself, not just symptoms. Here’s what Canadians need to know. - White, Brown, or Omega-3? Decoding The Egg Aisle
 Is a $4 carton better than a $10 one? We dive into CFIA regulations to reveal which eggs are health powerhouses and which are just hype.
Is a $4 carton better than a $10 one? We dive into CFIA regulations to reveal which eggs are health powerhouses and which are just hype.
The information provided on TheHealthInsider.ca is for educational purposes only and does not substitute for professional medical advice. TheHealthInsider.ca advises consulting a medical professional or healthcare provider when seeking medical advice, diagnoses, or treatment. To read about our editorial review process click here.