Summary
On January 26, 2026, Health Canada authorized Gazyva® to treat lupus nephritis, a serious kidney complication. While a single 1,000 mg vial costs approximately $5,477.84, the drug offers a powerful new way to achieve complete kidney recovery and is administered only twice a year after the first year of treatment.
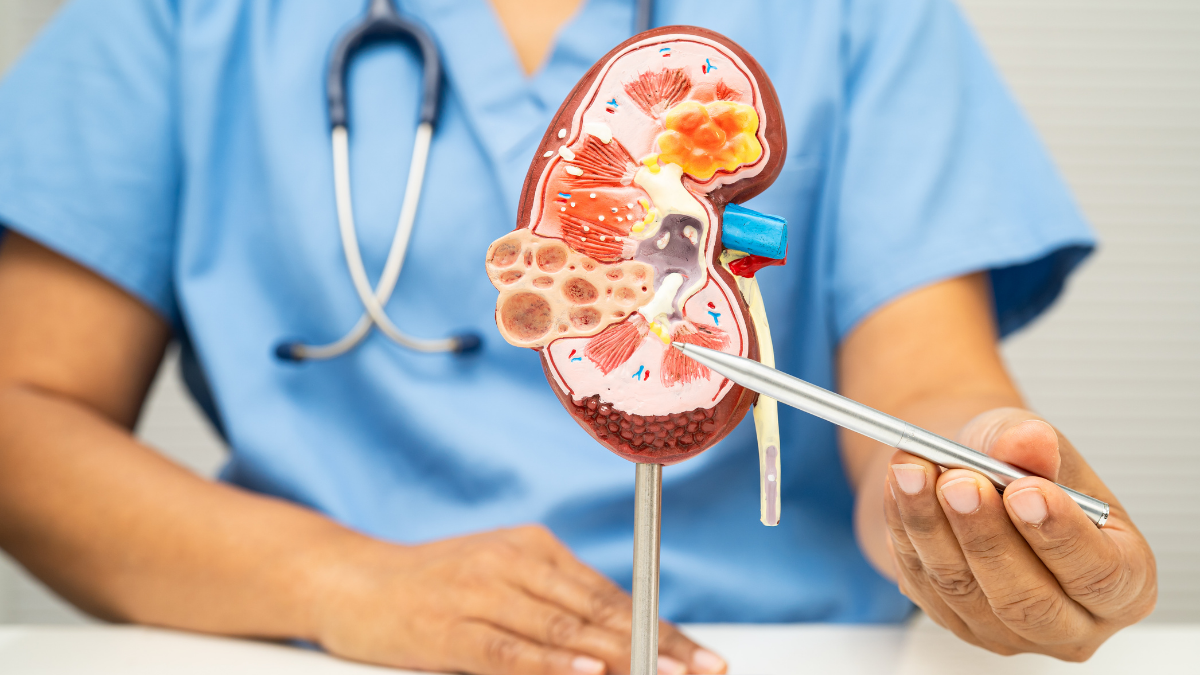
In a significant move for the autoimmune community, Health Canada has authorized Gazyva® (obinutuzumab) for adults with active lupus nephritis. Announced on January 26, 2026, this approval introduces the first and only monoclonal antibody shown to significantly improve “complete renal response” (a critical measure of kidney health) in patients facing this debilitating condition.
Lupus nephritis is a severe complication of Systemic Lupus Erythematosus (SLE), occurring when the immune system mistakenly attacks the kidneys. Without effective intervention, the condition causes irreversible damage to the kidney’s filtering structures (nephrons), often leading to kidney failure.
A New Standard of Care
The authorization is a “testament to the vision of bringing healthcare innovation to Canadians,” according to Roche Canada. For the 40% of lupus patients who develop kidney complications, Gazyva offers a way to potentially delay or even prevent the need for dialysis or a kidney transplant.
Unlike traditional therapies that may require frequent dosing, Gazyva follows a unique schedule:
- Initial Phase: Four doses administered within the first year.
- Maintenance Phase: Only two doses per year thereafter.
This twice-yearly maintenance schedule offers a significant convenience advantage over other targeted therapies currently on the market.
Coverage and Cost
As a high-specialty biologic, Gazyva represents a significant investment in long-term kidney preservation. According to CADTH reimbursement reviews, the wholesale price for a 1,000 mg vial of Gazyva is approximately USD$5,477.84.
While the total cost of treatment depends on the specific dosing cycle required for lupus nephritis (which includes an intensive first year followed by twice-yearly maintenance), the medication is often supported through:
- Provincial Health Plans: Roche Canada is currently working to secure listing on provincial formularies.
- Private Insurance: Many employer-based plans cover biologic treatments for chronic autoimmune conditions.
- Patient Support: Programs are typically available to help eligible patients navigate the high cost of specialty biologics and ensure access to treatment.
The Science: The REGENCY Study
The approval was driven by results from the Phase III REGENCY study, which tested Gazyva alongside standard therapies (such as corticosteroids and mycophenolate mofetil). The data was compelling:
- Higher Response Rates: Nearly 46.4% of patients treated with Gazyva achieved a complete renal response, compared to just 33.1% of those on standard therapy alone.
- Steroid Reduction: Patients were able to achieve better disease control while reducing their use of corticosteroids, which are often associated with harsh long-term side effects.
- B-Cell Targeting: Gazyva works by attaching to the CD20 protein on specific B-cells, depleting the “disease-causing” cells that drive the inflammation damaging the kidneys.
“Lupus nephritis is one of the most frequent, severe, and debilitating complications… treatment options are unfortunately limited,” said Dr. Ann Clarke of the University of Calgary. “Health Canada’s approval represents a significant advancement… offering renewed hope.”
Access and Availability
Lupus affects approximately 1 in 1,000 Canadians, predominantly women of childbearing age and people of colour. Because the disease is complex and unpredictable, expanded access to evidence-based therapies is vital.
Roche Canada has stated they are committed to working with provincial and territorial jurisdictions to ensure Gazyva is added to public and private drug plans as soon as possible.
Subscribe to The Health Insider newsletter where you’ll find many more insights – delivered straight to your inbox.
~ Read more from The Health Insider ~
- Ditching your Sunscreen? Separate Fact From FictionTrading sunscreen for internet rumours exposes your skin to a proven Class 1 carcinogen — the sun. Read the risk analysis and learn about new options before heading outside.
- Would You Trust AI Over Your Doctor?Nearly half of Canadians use AI for medical advice, but 68% would still wait two weeks for a human doctor over an immediate AI diagnosis.
- Syphilis Is Surging in Ontario and Pregnant Women Are Paying the PriceOntario’s syphilis surge has reached every corner of the province. Here’s what pregnant people need to know about screening and protection.
- Red Light Therapy: Separating Science From HypeBeyond the wellness trend lies real clinical proof. Discover how red light therapy physically heals cells, targets chronic pain, and regrows hair.
- Beyond Ibuprofen: New Tech and Natural Remedies for Period PainPeriod pain isn’t just something you have to endure. Discover the natural remedies and Health Canada-approved tech that offer proven relief.
- PCOS Has a New Name and This is Why it MattersPCOS is now PMOS (Polyendocrine Metabolic Ovarian Syndrome). Discover why this landmark renaming matters for patient diagnosis and metabolic care.
The information provided on TheHealthInsider.ca is for educational purposes only and does not substitute for professional medical advice. TheHealthInsider.ca advises consulting a medical professional or healthcare provider when seeking medical advice, diagnoses, or treatment. To read about our editorial review process click here.