Summary
Health Canada authorized Nemluvio®, a first-in-class biologic for eczema and prurigo nodularis that specifically targets the biological cause of chronic itch. While national funding reviews are still underway, this approval provides a vital new option for patients who haven't found relief with standard creams.
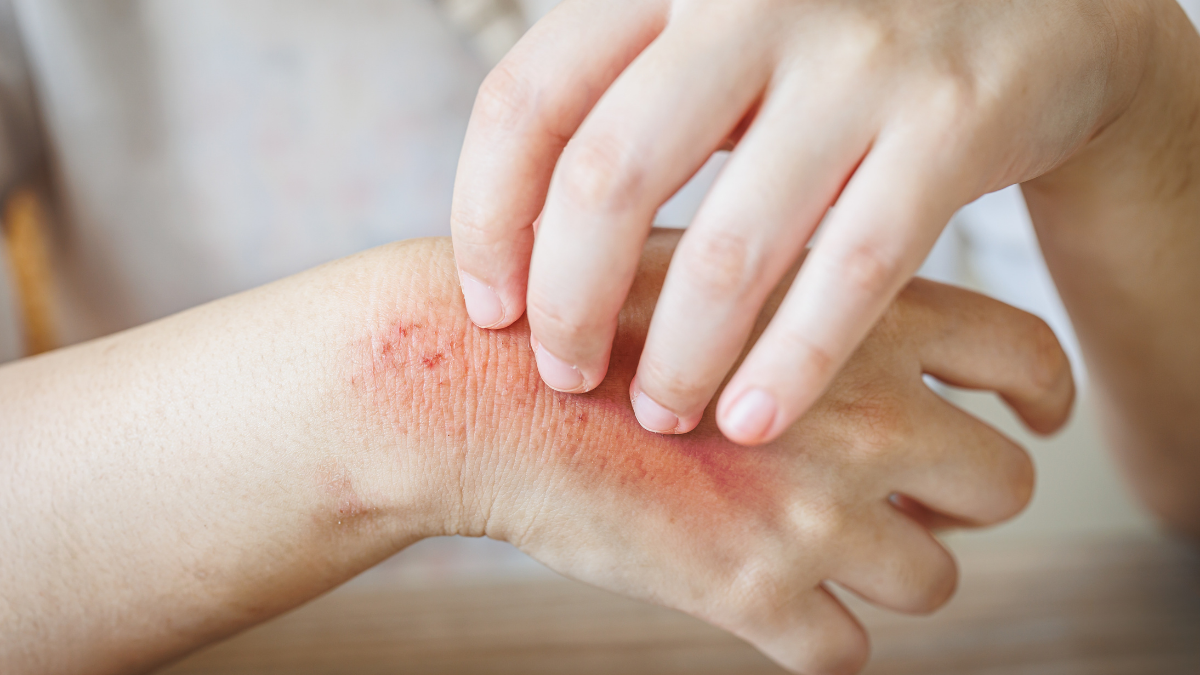
A new era in dermatological care has arrived for Canadians suffering from chronic, debilitating itch. Health Canada authorized Nemluvio® (nemolizumab) for the treatment of moderate-to-severe atopic dermatitis (eczema) in patients aged 12 and older, as well as prurigo nodularis in adults.
Nemluvio® is a first-in-class biologic that specifically targets the neuro-immune pathway responsible for itch, providing a much-needed alternative for patients whose conditions are not adequately controlled by topical therapies.
Targeting the “Itch Cytokine”
Unlike traditional eczema treatments that focus primarily on skin inflammation, Nemluvio® is the first monoclonal antibody designed to block interleukin-31 (IL-31). Often referred to by scientists as the “itch cytokine,” IL-31 sends signals directly to the nervous system to trigger the urge to scratch.
By blocking this signal, Nemluvio® addresses the most burdensome symptom of these conditions: the relentless itch that leads to sleep deprivation, skin damage, and significant mental health strain.
“Nemolizumab’s ability to rapidly target itch makes it an exciting and much-needed advancement, offering patients relief where they need it most,” said Dr. Chih-ho Hong, Board-Certified Dermatologist, MD, FRCPC, Surrey, BC.
Proven Clinical Success
The Health Canada authorization was based on robust data from two major Phase III clinical programs:
- ARCADIA 1 & 2 (Atopic Dermatitis): These trials demonstrated that Nemluvio, administered every four weeks, significantly improved skin lesions and reduced itch in adults and adolescents.
- OLYMPIA 1 & 2 (Prurigo Nodularis): This program, the largest of its kind for this condition, showed that the treatment significantly improved itch and cleared skin nodules compared to a placebo.
Status in Canada: The Road to Access
While Nemluvio® is now authorized for sale in Canada, its widespread availability under public insurance is still in progress.
- Reimbursement Review: The drug is currently undergoing a cost-effectiveness review by Canada’s Drug Agency (CDA-AMC). This national review is a required step before the drug can be listed on federal, provincial, and territorial public drug plans.
- Cost & Funding: Because the review is ongoing, an official, widely available list price for patient use has not yet been finalized or publicly released. Until funding decisions are made by the provinces, Nemluvio® is not yet a regular benefit under most public insurance plans.
- Patient Support: Historically, specialty biologics are supported by manufacturer-led patient assistance programs. These programs often help patients navigate early access and insurance coverage during the reimbursement review phase.
Is It Right For You?
Nemluvio® is intended for those whose disease is not adequately controlled with topical prescription therapies. Because it is an injectable medication given once every four weeks, it offers a convenient schedule for those looking to move away from daily creams or more frequent injections.
Subscribe to The Health Insider newsletter for more evidence-based updates on Canadian medical breakthroughs.
~ Read more from The Health Insider ~
- Save on Ozempic: Generic Swaps Arrive in CanadaGeneric Ozempic has arrived in Canada, slashing costs by two-thirds. Learn how this historic price drop benefits your health and wallet.
- Nine in Ten Canadians Want the Health System to Change. Here’s What They’re Saying.A new national survey finds 91% of Canadians want health system change now, with broad openness to team-based care, digital tools, and private delivery.
- Cataract Awareness: Guard Your Eyes from UV DamageJune is Cataract Awareness Month. Learn how wearing 100% UV blocking sunglasses protects your eyes and reduces your risk of cataracts over time.
- Your Social Media Feed May Be Selling You Women’s Health Products That Don’t WorkWomen’s health gaps are being filled by viral marketing. Here is the clinical reality behind 5 heavily promoted online wellness trends.
- Ditching your Sunscreen? Separate Fact From FictionTrading sunscreen for internet rumours exposes your skin to a proven Class 1 carcinogen — the sun. Read the risk analysis and learn about new options before heading outside.
- Would You Trust AI Over Your Doctor?Nearly half of Canadians use AI for medical advice, but 68% would still wait two weeks for a human doctor over an immediate AI diagnosis.
The information provided on TheHealthInsider.ca is for educational purposes only and does not substitute for professional medical advice. TheHealthInsider.ca advises consulting a medical professional or healthcare provider when seeking medical advice, diagnoses, or treatment. To read about our editorial review process click here.